
Home / Publications / Journals / Nuclear Technology / Volume 130 / Number 2
Radioactive Waste Minimization by Electrolytic Extraction and Destruction in a Purex-Truex Actinide Separation System
Nuclear Technology / Volume 130 / Number 2 / May 2000 / Pages 196-205
Technical Paper / Radioactive Waste Management and Disposal / dx.doi.org/10.13182/NT00-A3087
Articles are hosted by Taylor and Francis Online.
Electrolytic extraction of noble metals from nitric acid media was investigated. The largest deposition yield was obtained for Pd, supported by its large rate constants. Rate constants of RuNO3+ and ReO4- were, however, smaller than that of Pd2+; their yield can be improved under high cathode current supply in lower nitric acid concentration. Rather high apparent activation energy was observed for the deposition of RuNO3+. Peculiar masking or synergistic effects in their electrodeposition behaviors might be due to mutual interaction of RuNO3+, Pd2+ with ReO4- in nitric acid solution. Sufficiently different redissolution potentials for deposited metals indicate their fractional recovery by anode processing.
Mediatory electrochemical oxidation (MEO) was investigated for the mineralization of waste O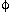 D[iB]CMPO (hereafter CMPO) by burning its bulky hydrocarbon moiety under the existence of various kinds of metal ions. Only Ag2+/+ offered high-current efficiency up to 75%, fairly exceeding that by direct electrooxidation. Redox coupling characterized by a simple electron transfer, Mm+ + ne- <=> M(m-n)+ provided high E0, will act exactly as an active mediator. As for the destruction paths for CMPO by MEO, cleavage between carbonyl C and N of amide moiety was of principal importance. The coupling of Co3+/2+ is also recommended because of hydraulic advantages.
D[iB]CMPO (hereafter CMPO) by burning its bulky hydrocarbon moiety under the existence of various kinds of metal ions. Only Ag2+/+ offered high-current efficiency up to 75%, fairly exceeding that by direct electrooxidation. Redox coupling characterized by a simple electron transfer, Mm+ + ne- <=> M(m-n)+ provided high E0, will act exactly as an active mediator. As for the destruction paths for CMPO by MEO, cleavage between carbonyl C and N of amide moiety was of principal importance. The coupling of Co3+/2+ is also recommended because of hydraulic advantages.

