The Synergistic Effects in Organophosphate Extraction Systems
Nuclear Science and Engineering / Volume 16 / Number 4 / August 1963 / Pages 405-412
Technical Paper / dx.doi.org/10.13182/NSE63-A26552
Articles are hosted by Taylor and Francis Online.
Combinations of dialkylphosphoric acids (HA) and neutral organophosphorus reagents (B) have for some time been known to produce enhanced (synergistic) extraction of 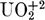 and other
and other 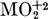 actinide ions. This effect may be attributed to the formation of a mixed complex—either a substitution product MO2A3HB or an addition product MO2A4H2B resulting from the reaction of B with the extraction complex MO2A4H2 normally formed with HA alone. Available extraction data, when analyzed in terms of the competing hydrogen-bonding interactions between HA and B, appear more consistent with an addition product MO2A4H2B. The stability of this mixed complex as a function of the neutral organophosphate used and as a function of the diluent used is consistent with hydrogen bonding or solvation of MO2A4H2 by B. The much stronger and more general synergism found in systems wherein HA is replaced by the β-diketone, Thenoyltrifluoroacetone (HY) may be attributed to the relatively weaker competing interaction between HY and B and to the less coordinatively saturated extraction complexes formed by HY alone.
actinide ions. This effect may be attributed to the formation of a mixed complex—either a substitution product MO2A3HB or an addition product MO2A4H2B resulting from the reaction of B with the extraction complex MO2A4H2 normally formed with HA alone. Available extraction data, when analyzed in terms of the competing hydrogen-bonding interactions between HA and B, appear more consistent with an addition product MO2A4H2B. The stability of this mixed complex as a function of the neutral organophosphate used and as a function of the diluent used is consistent with hydrogen bonding or solvation of MO2A4H2 by B. The much stronger and more general synergism found in systems wherein HA is replaced by the β-diketone, Thenoyltrifluoroacetone (HY) may be attributed to the relatively weaker competing interaction between HY and B and to the less coordinatively saturated extraction complexes formed by HY alone.


